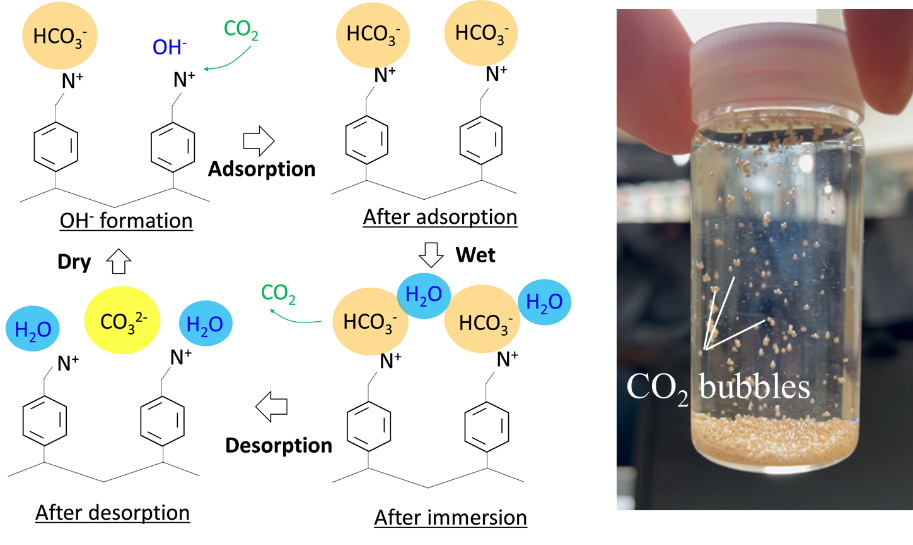
CO2 Capture by Moisture Swing Adsorption
In recent years, efforts have been made to capture, store, and utilize CO2 from exhaust gases and the atmosphere. A key element in these efforts is the development of effective CO2 capture technologies. Various approaches, such as physical adsorption and chemical absorption, have been proposed; however, reducing energy consumption remains a major challenge. To address this issue, this study focuses on moisture swing adsorption, in which CO2 is adsorbed when the sorbent is dry and released when it becomes humid. In this method, CO2 can be desorbed simply by supplying water, offering the potential for a significant reduction in energy consumption compared with conventional thermally driven CO2 capture systems. This research promotes an integrated approach spanning chemistry and mechanical engineering, covering the development of polymer-based sorbents, the construction of simulation models, and the design and development of the overall system.
(1)Development of Adsorbent Materials
High adsorption capacity and rapid adsorption kinetics are required for the adsorbent. To achieve this, novel polymer structures are being developed. Molecular dynamics simulations are employed to investigate the distribution of water within the polymer and the diffusion behavior of CO2, enabling the exploration of chemical structures suitable for moisture swing adsorption. In addition, efforts are being made to fabricate the adsorbent into porous forms, achieving both high adsorption performance and practical applicability in real systems. Accordingly, high-performance CO2 adsorbents are being developed through the combined optimization of polymer structures and the design of porous morphologies. At present, a small-scale but continuous production capability is being established.

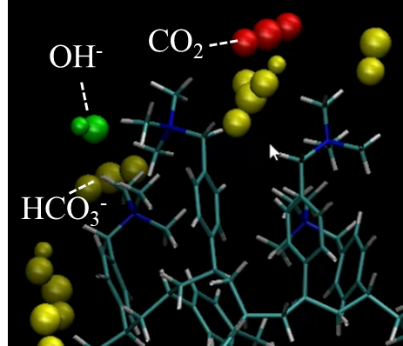
(2)Elucidation of Mass Transport Mechanisms
To develop high-performance adsorbents, it is essential to elucidate the transport mechanisms of CO2 and other species within the adsorbent. To this end, techniques for visualizing CO2 within polymers are being developed, enabling investigation of the relationship between CO2 transport mechanisms and chemical structure. In addition, based on the insights obtained, a three-dimensional simulation model capable of reproducing adsorption and desorption processes is under development, promoting research that bridges phenomena from the nanoscale to the meter scale.
(3)System Development for Moisture Swing Adsorption
Reducing energy consumption requires effective energy management of the CO2 capture system. Accordingly, a system capable of capturing approximately 1.0 t/year of CO2 from ambient air has been developed. Based on the data obtained, efforts are underway to establish the optimal system configuration and operating conditions that minimize energy consumption. A prototype unit has already been completed, and demonstration experiments have been initiated. Moving forward, a process simulation model will be constructed based on the acquired data to further refine and establish optimal operating modes.
CO2 Electrochemical Conversion to Organic Compounds
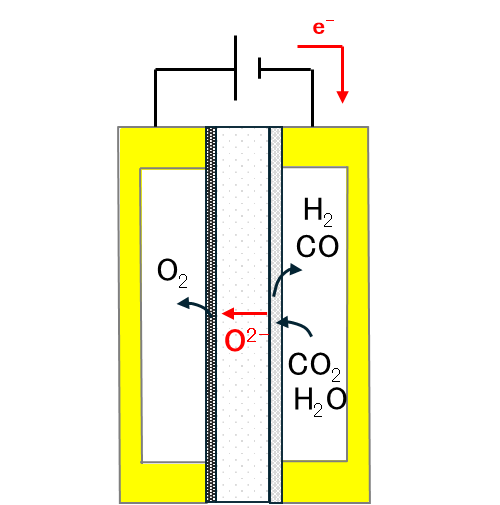
Research is being conducted on electrochemical technologies to efficiently convert CO2 captured by moisture swing adsorption into CO, a valuable feedstock for organic synthesis. In particular, solid oxide electrolysis cells (SOECs) are the primary focus, with efforts directed toward performance optimization based on differences in reaction mechanisms and transport characteristics. The effects of moisture and trace impurities (e.g., oxygen, nitrogen, and volatile organic compounds) contained in the captured CO2 on electrode reactions, catalyst degradation, and ionic conductivity are being systematically evaluated to clarify stable operating regimes under practical conditions. In parallel, based on the analysis of degradation factors during long-term operation, design guidelines for improving cell durability are being established, along with optimization of the overall system, including gas supply and product separation. In addition, cell configurations that enable effective utilization of oxygen generated during CO2 electrolysis are also being investigated. Through these efforts, the aim is to contribute to the valorization of captured CO2 and the realization of sustainable chemical processes.
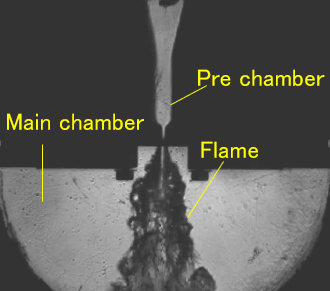
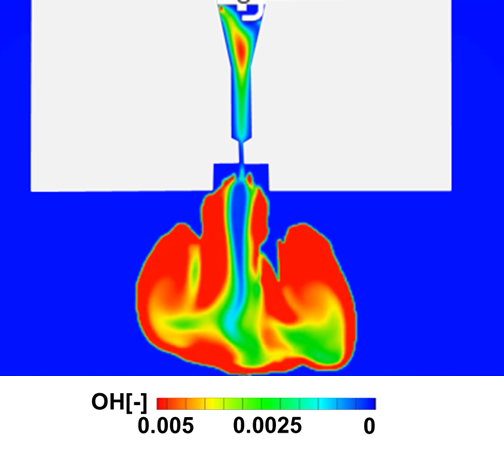
Pre-chamber Jet Combustion Suitable for Synthetic Fuels
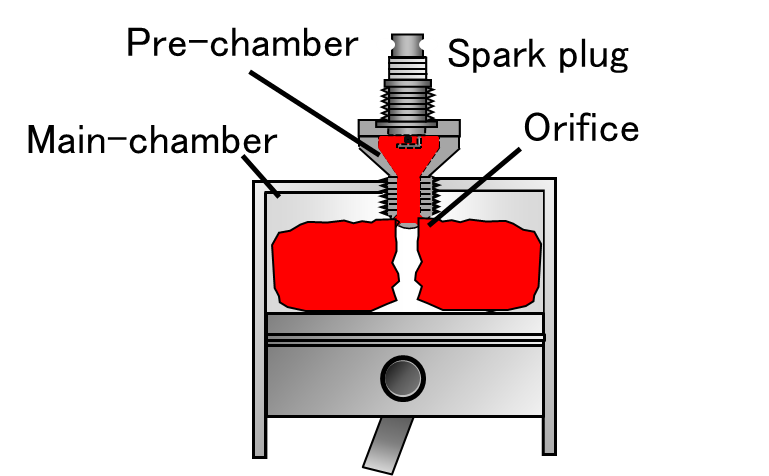
Research is being conducted on pre-chamber jet combustion, a combustion technology that enables efficient utilization of synthetic fuels derived from CO2. In this approach, the fuel in the pre-chamber is ignited and combusted to generate high-temperature, high-velocity flame jets that are injected into the main chamber, achieving distributed multi-point ignition over a wide region. This leads to a shortened combustion duration and contributes to improved engine efficiency. In pre-chamber jet combustion, the geometry of the orifice connecting the pre-chamber and the main chamber strongly influences jet structure, turbulence intensity, and flame propagation behavior, making it a critical factor governing combustion stability. To address this, optical diagnostics are employed to visualize the combustion process from the pre-chamber to the main chamber in detail, while numerical simulations incorporating chemical kinetics are combined to elucidate the underlying mechanisms of pre-chamber jet combustion using synthetic fuels. Furthermore, toward establishing design guidelines for fuels suitable for pre-chamber jet combustion, attention is given to dimensionless parameters such as the Lewis number, and their relationships with ignitability and combustion duration are systematically evaluated. These efforts aim to enable the rational design of synthetic fuels optimized for pre-chamber jet combustion.
Polymer Electrolyte Membrane Fuel Cell (PEMFC)
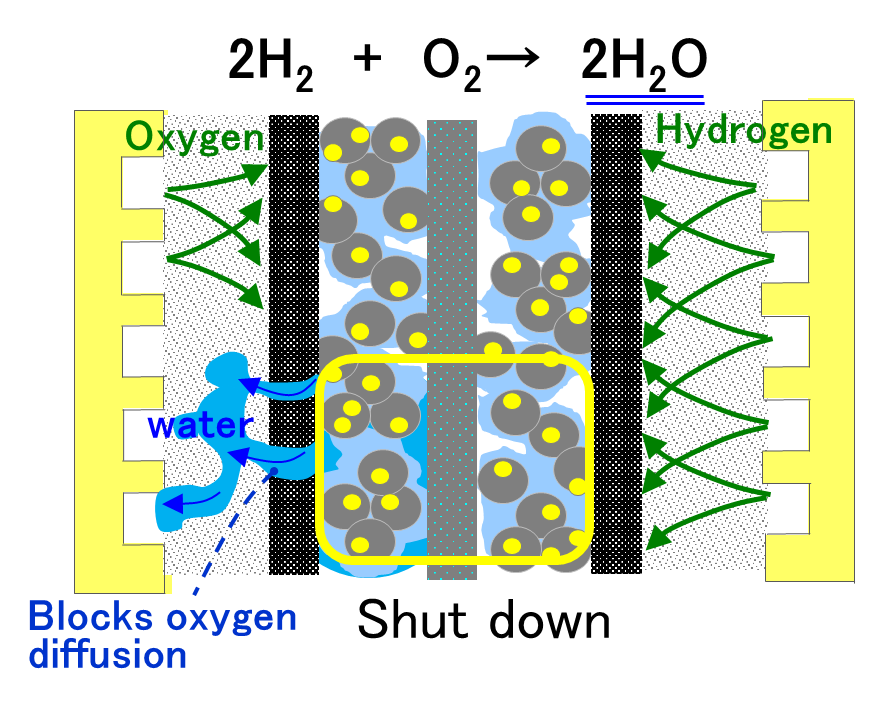
Focusing on polymer electrolyte fuel cells (PEFCs), which serve as core energy conversion devices in decarbonization technologies enabling highly efficient utilization of hydrogen, this research investigates and controls water and oxygen transport phenomena within the cell. In PEFCs, electricity is generated through electrochemical reactions between hydrogen and oxygen, with water produced as a byproduct. While this generated water contributes to maintaining membrane hydration, excessive accumulation can impede oxygen transport within the gas diffusion layer and catalyst layer, leading to flooding and a consequent deterioration in performance.
To address these challenges, this study focuses on multiphase flow and mass transport characteristics in the porous structures inside the cell, aiming to establish design guidelines that simultaneously ensure efficient water removal and sufficient oxygen supply. In particular, an approach is being pursued in which the gas diffusion layer (GDL) is thinned to the limit to reduce diffusion resistance, thereby enhancing oxygen transport while promoting rapid water removal.
Furthermore, a numerical simulation framework based on the lattice Boltzmann method (LBM) has been developed to reproduce complex transport phenomena of water and oxygen within porous media at high resolution, which are difficult to capture using conventional continuum models. This enables quantitative clarification of the relationship between microscale transport phenomena and cell performance, contributing to the optimization of structural design and operating conditions.
(1)Two-Phase Flow Simulation Using the Lattice Boltzmann Method
Because polymer electrolyte fuel cells contain fine porous structures, two-phase flow simulations have conventionally suffered from extremely high computational cost. To address this issue, a lattice Boltzmann method (LBM), which is well suited for parallel computing, has been employed. In addition, a stable numerical scheme capable of handling relatively large time steps has been developed. Computational cost has further been reduced by introducing approaches such as the multi-block method, in which spatial resolution is selectively coarsened in non-critical regions.
Currently, the model is being extended to incorporate water phase change as well as oxygen transport, while simultaneously advancing model development and further reduction of computational cost.
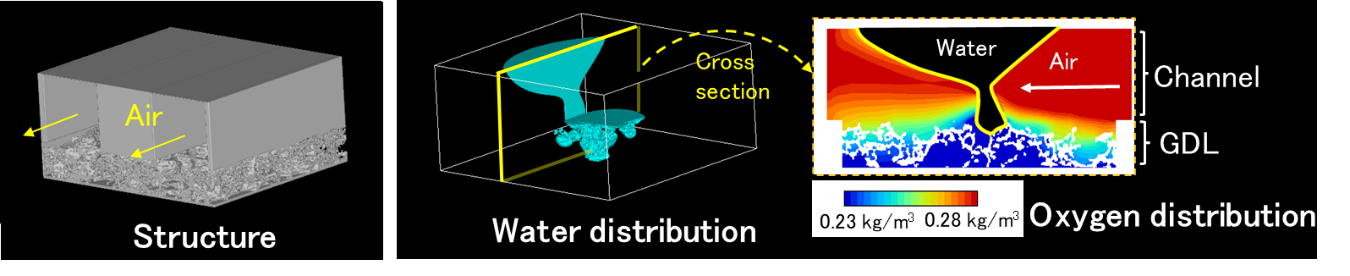
(2)Gas Diffusion Layer / Microporous Structure

In polymer electrolyte fuel cells, reducing oxygen transport resistance on the cathode side is directly linked to performance improvement in high current density regions. To address this challenge, a strategy focusing on reducing the thickness of the gas diffusion layer (GDL) has been adopted, leading to the development of an ultra-thin GDL with a thickness of less than 40 µm and precisely controlled pore size distribution. As a result, diffusion resistance has been significantly reduced, achieving approximately a twofold improvement in oxygen transport performance compared with conventional designs.
Furthermore, to deepen the understanding of transport phenomena within the GDL, a measurement technique using infrared imaging has been developed to visualize micrometer-scale pore structures and water distribution. This approach enables high-spatial-resolution observation of gas and liquid water distribution under operating conditions, contributing to the elucidation of transport-dominant mechanisms and the advancement of material design guidelines.
